Regulatory Information Management Market to Hit USD 7.6 Billion by 2036, Led by US, China with Veeva & ArisGlobal
Rising regulatory complexity and digital compliance adoption drive RIM market growth to USD 7.6B by 2036, led by cloud platforms and pharma demand.
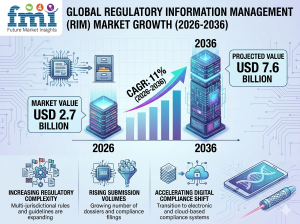
NEWARK, DE, UNITED STATES, March 30, 2026 /EINPresswire.com/ -- The global Regulatory Information Management (RIM) Market is valued at USD 2.7 billion in 2026 and is projected to reach USD 7.6 billion by 2036, expanding at a CAGR of 11% from 2026 to 2036. Growth is driven by increasing regulatory complexity, rising submission volumes, and the accelerating shift toward digital compliance systems across pharmaceutical, biotech, and medical device industries.
As global regulatory frameworks become more stringent and interconnected, organizations are transitioning from manual processes to centralized digital platforms. Regulatory information management solutions are emerging as critical infrastructure for ensuring compliance, improving data accuracy, and accelerating product approvals across multiple markets.
Request For Sample Report | Customize Report | Purchase Full Report
https://www.futuremarketinsights.com/reports/sample/rep-gb-1889
Regulatory Information Management Market Snapshot (2026–2036)
• Market size in 2026: USD 2.7 billion
• Market size in 2036: USD 7.6 billion
• CAGR (2026–2036): 11%
• Leading segment: Software solutions (~70% share)
• Top end-use segment: Pharmaceuticals (45% share)
• Dominant deployment: Cloud-based platforms
• Fastest-growing country: India
• Key companies: Veeva Systems, ArisGlobal, PhlexGlobal, Amplexor Life Sciences, MasterControl
Market Momentum
The RIM market begins at USD 2.7 billion in 2026, supported by strong demand from pharmaceutical companies managing increasing global submission requirements. Between 2027 and 2030, adoption accelerates as organizations invest in cloud-based platforms to streamline regulatory workflows and improve operational efficiency.
Entering 2032 and beyond, integration of artificial intelligence, automation, and analytics transforms regulatory processes, enabling faster approvals and improved lifecycle management. By 2036, the market reaches USD 7.6 billion, driven by widespread digital transformation across life sciences and increasing reliance on centralized compliance systems.
Why the Market is Growing
The Regulatory Information Management Market is expanding as life sciences organizations face mounting pressure to comply with evolving global regulations while maintaining speed and efficiency. Increasing submission volumes, frequent product updates, and multi-country compliance requirements are driving demand for centralized systems that ensure consistency and accuracy.
Cloud-based RIM platforms are gaining strong traction due to their scalability, real-time collaboration capabilities, and ability to support globally distributed regulatory teams. These solutions reduce manual workloads, improve document traceability, and enable faster response to regulatory changes.
Additionally, the rise of biologics, advanced therapies, and digital health products is increasing documentation complexity, further reinforcing the need for structured regulatory systems. Companies are prioritizing digital transformation strategies to enhance compliance confidence and accelerate time-to-market.
Segment Spotlight
1. Solution: Software Dominates Market Demand
Software solutions account for approximately 70% of the global market, driven by their ability to manage submissions, registrations, labeling, and lifecycle tracking. Cloud-based platforms contribute nearly 60% within this segment, offering flexibility and scalability.
2. Deployment: Cloud-Based Platforms Lead Adoption
Cloud deployment is becoming the preferred choice due to faster implementation, lower upfront costs, and enhanced accessibility across global teams. On-premise systems continue to serve organizations with strict data governance requirements.
3. Vertical: Pharmaceuticals Hold Largest Share
Pharmaceutical companies represent around 45% of total demand, supported by high submission frequency, strict regulatory scrutiny, and complex lifecycle management requirements.
Drivers, Opportunities, Trends, Challenges
• Drivers: Rising regulatory complexity, increased submission volumes, digital transformation initiatives
• Opportunities: AI-driven regulatory workflows, cloud adoption, global harmonization standards
• Trends: Shift toward centralized platforms, automation in submissions, real-time compliance tracking
• Challenges: High implementation costs, data security concerns, regulatory variability across regions
Country Growth Outlook (2026–2036)
India leads the market with a CAGR of 12.8%, driven by rapid pharmaceutical exports and regulatory digitization initiatives. China follows at 12.4%, supported by regulatory reforms and increased global alignment. Japan (11.5%) benefits from harmonization with international standards, while Germany (11.2%) and the United Kingdom (10.8%) see steady growth due to evolving compliance frameworks. The United States grows at 10.3%, supported by high submission volumes and advanced regulatory requirements.
Competitive Landscape
The Regulatory Information Management Market is moderately consolidated, with competition centered on platform scalability, regulatory expertise, and digital capabilities. Leading players are focusing on cloud-native solutions, integration capabilities, and automation to enhance regulatory efficiency.
Companies are investing in advanced technologies such as artificial intelligence and analytics to improve submission accuracy and reduce approval timelines. Strategic priorities include expanding global compliance capabilities, strengthening client relationships, and enhancing data security frameworks.
Vendors are also differentiating through hybrid deployment models, consulting services, and tailored solutions for small and medium enterprises. As regulatory demands intensify, the market increasingly favors providers that deliver both technological innovation and deep regulatory expertise.
Frequently Asked Questions (FAQ)
What is the global Regulatory Information Management Market size?
The market is valued at USD 2.7 billion in 2026 and is projected to reach USD 7.6 billion by 2036.
At what rate is the market expected to grow?
The market is expected to expand at a CAGR of 11% from 2026 to 2036.
What is regulatory information management?
It refers to digital systems used to manage regulatory submissions, compliance data, labeling, and product lifecycle processes across global markets.
Why is RIM adoption increasing?
Growing regulatory complexity, digital transformation, and the need for faster approvals are driving widespread adoption.
Explore More Related Studies Published by FMI Research
Internet of Everything (IoE) Market - https://www.futuremarketinsights.com/reports/global-internet-of-everything-market
Modular Data Center Market - https://www.futuremarketinsights.com/reports/modular-data-center-market
Digital Forensics Market - https://www.futuremarketinsights.com/reports/digital-forensics-market
About Future Market Insights (FMI)
Future Market Insights, Inc. (FMI) is an ESOMAR-certified, ISO 9001:2015 market research and consulting organization, trusted by Fortune 500 companies and global enterprises. With a strong presence across the United States, United Kingdom, India, and the Middle East, FMI delivers data-driven insights across 30+ industries and over 1200 markets worldwide.
Why Choose FMI:
https://www.futuremarketinsights.com/why-fmi
Sudip Saha
Future Market Insights Inc.
+1 347-918-3531
rahul.singh@futuremarketinsights.com
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.